|
The electron configuration of sodium shows that the last shell. Valence electrons are the electrons present in the outermost shell of an atom. The total number of electrons in a valence shell is called a valence electron. The last shell after the electron configuration is called the valence shell. The third step is to diagnose the valence shell (orbit). Look at the group that the element is in, as the group number. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. The alkaline earths tend to display a valence of 2. Step-3: Determine the valence shell and calculate the total electrons. On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. The alkali metals, for example, almost always display a valence of 1. Most elements have preferred values of valence electrons. This electron configuration of vanadium ion (V 3+) shows that vanadium ion has three shells and the last shell has ten electrons. 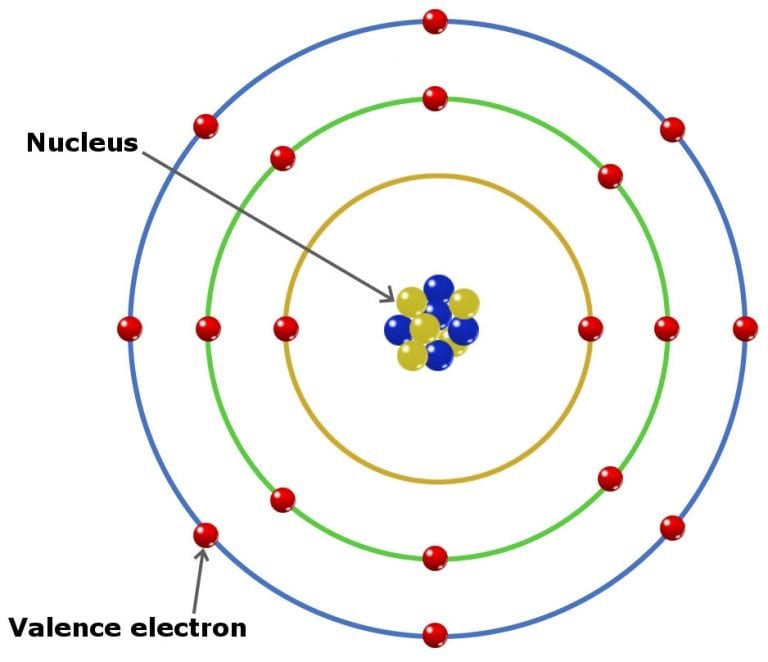
On the other hand, The electron configuration of vanadium ions (V 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 2. 
\) experienced by the valence electrons in the magnesium anion, the neutral magnesium atom, and magnesium cation? Use the simple approximation for shielding constants. However, in practical use, main group elements of the periodic table may display any valence from 1 to 7 (since 8 is a complete octet). For this, the valence electrons of the vanadium ion (V 2+) are eleven.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
